In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
By A Mystery Man Writer

In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state
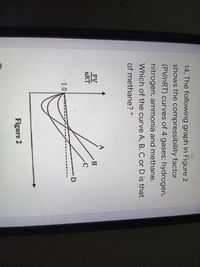
Answered: 14, The following graph in Figure 2…

Compressibility factor - Wikipedia
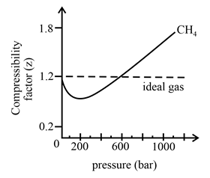
In the following compressibility factor Zvs pressure graph at 300Kthe compressibility of CH4 at pressures 200bardeviates from ideal behaviour because

Energies, Free Full-Text

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

The graph of compressibility factor Z vs P for one mole of a real gas is shown in following diagram. The graph is plotted at a constant temperature 273 K. If the

Real-gas z-factor, as attributed to Standing and Katz, 9 plotted as a

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

Compressibility Factor Calculator

As the pressure approaching zero i.e., very low pressure, the curves plotted between compressibility factor Z and P n mole of gases have the following characteristics.I. The intercept on the y-axis leads

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

Compressibility Factor Z Important Concepts and Tips for JEE Main
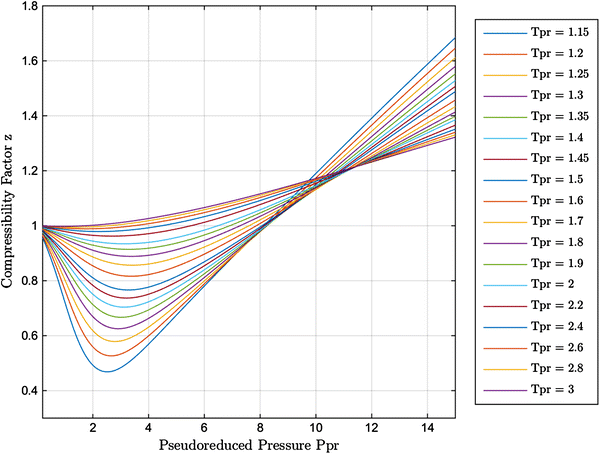
New explicit correlation for the compressibility factor of natural gas: linearized z-factor isotherms

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download