By A Mystery Man Writer

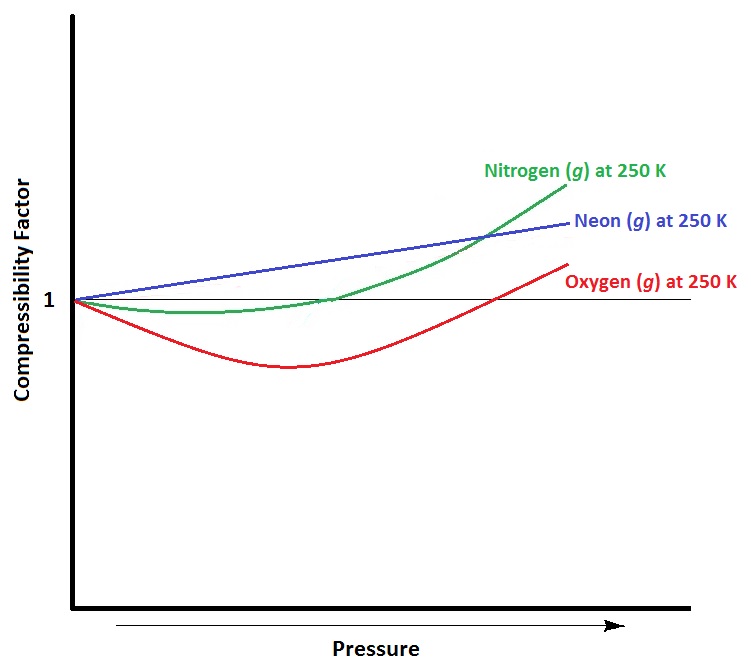
By the graph, at T2, the gas does not deviate from ideal gas behaviour at low pressure (Z = 1).Hence, T2 = TBT2 = TB (Boyle's temperature)T1 > TB (Here, Z > 1)This implies that T3 B and TB > TC(Here, Z So, T1 and T2 must be above TC.
Real Gases – Introductory Chemistry – 1st Canadian Edition

As shown in the figure 200 K and 500 atm value of compressibility factor is 2 (approx). Then the volume of the gas this point will be:0.09 L0.065 L0.657 L0.01 L

physical chemistry - Compressibility Factor Graph - Which gas attains a deeper minimum? - Chemistry Stack Exchange

Compressibility factor - Wikipedia

Pressure versus volume graph for a real gas and an ideal gas are shown in figure. Answer the following question on the basis of this graph.i Interpret the behaviour of real gas

The graph of compressibility factor (Z) v/s P 1 mol of a real gas is shown in following diagram. The graph is plotted 273 K temperature. If slope of graph very high

Compressibility factor (gases) - Citizendium

Variation of both compressibility factor and viscosity of gas with
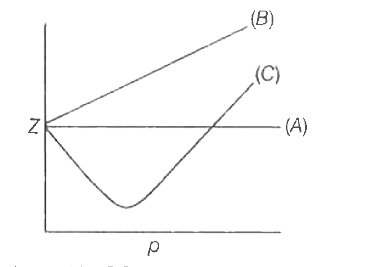
Telugu] The variation of compressibility factor (Z) with pressure (p

The height of a waterfall is 50 m. If g = 9.8 msandndash;2 the difference between the temperature at the top and the bottom of the waterfall is:a)1.17 anddeg;Cb)2.17anddeg; Cc)0.117anddeg; Cd)1.43anddeg; CCorrect

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

Plot of experimental measurements of the z-factor