In the following compressibility factor Z vs pressure graph at 300
By A Mystery Man Writer

In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

Gas Compressibility - an overview

Improved description of the liquid phase properties of Methane: density, enthalpy, plus saturated vapor compressibility factor

Solved 20+ 15 10 9. Consider the gas compressibility factor

Gas Laws / Gases Behaviour, PDF, Gases
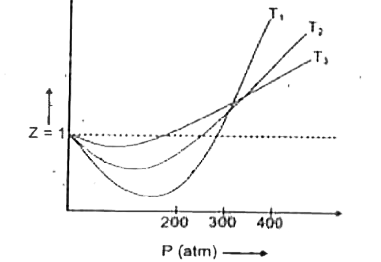
Compressibility factor (Z) is plotted against pressure at different te

Energies, Free Full-Text

If the slope of 'Z' (compressibility factor) vs. 'p' curve is constant

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download