In the following compressibility factor (Z) vs. pressure graph 300 K, the compressibility of CH_{4} pressure < 200 bar deviates from ideal behaviour becauseThe molar volume of CH_{4} is than its molar
By A Mystery Man Writer

Click here:point_up_2:to get an answer to your question :writing_hand:in the following compressibility factor z vs pressure graph at 300 k the compressibility of
Click here👆to get an answer to your question ✍️ In the following compressibility factor -Z- vs- pressure graph 300 K- the compressibility of CH-4- pressure - 200 bar deviates from ideal behaviour becauseThe molar volume of CH-4- is than its molar volume in the ideal stateThe molar volume of CH-4- is than its molar volume in the ideal stateThe molar volume of CH-4- is same as that in its ideal stateIntermolecular interactions between CH-4- molecules decreases

1.1: Thermodynamic Variables and Equations of State - Chemistry LibreTexts

Chemical Thermodynamics

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

Fundamentals SpringerLink
States of Matter Class 11 Notes CBSE Chemistry Chapter 5 [PDF]

Objectives_template

Compressibility factor - Wikipedia

Punjabi] The graph of compressibility factor (Z) vs. P for one mole o
Is z (compressibility factor) vs P (pressure) graph drawn by changing volume? If it is why it isn't drawn by changing mole - Quora
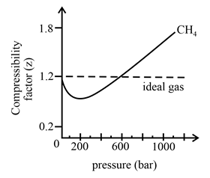
In the following compressibility factor Zvs pressure graph at 300Kthe compressibility of CH4 at pressures 200bardeviates from ideal behaviour because
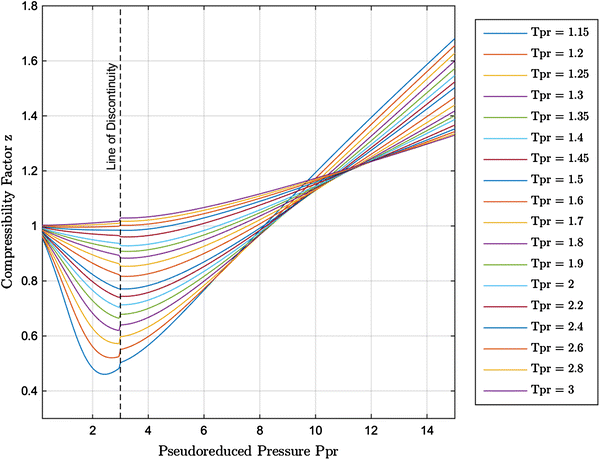
New explicit correlation for the compressibility factor of natural gas: linearized z-factor isotherms
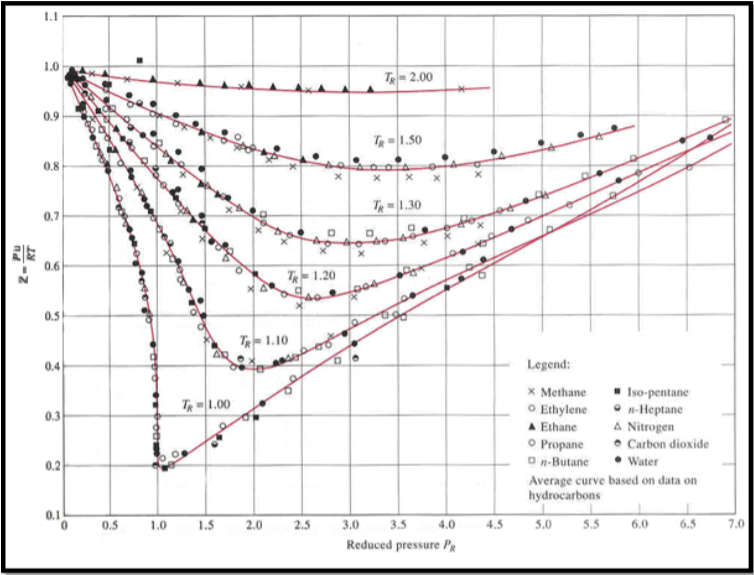
Solved Use the plot of compression factor (Z) vs reduced

4.2: Real Gases (Deviations From Ideal Behavior) - Chemistry LibreTexts

In the following compressibility factor (Z) vs. pressure graph 300 K, the compressibility of CH_{4} pressure < 200 bar deviates from ideal behaviour becauseThe molar volume of CH_{4} is than its molar
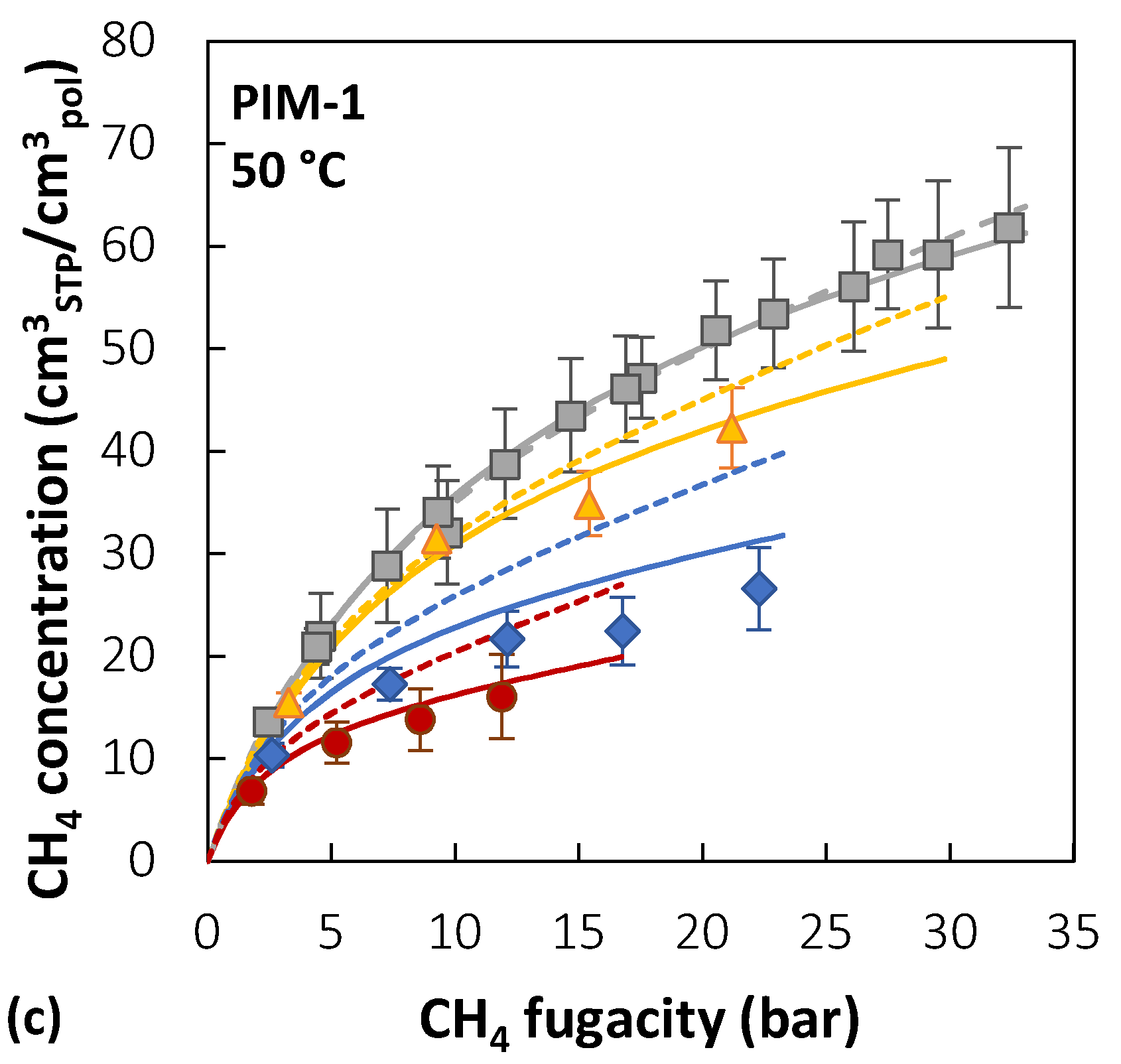
Membranes, Free Full-Text