
How many moles of methane are required to produce 22g CO2 (g) after combustion? #ncert #11

i.ytimg.com/vi/EjICJZdDcRo/hq720.jpg?sqp=-oaymwEhC
How many water molecules will be formed from the combustion of 5 atoms of oxygen and whatever amount of methane with balanced equation? - Quora
How many molecules are present in 2.24 ml of CO2 measured at STP? - Quora

How many moles of methane are required to produce `22 g CO_(2)` for combustion ?
When 100 gm of CaCO3 is heated, what amount of CO2 is formed? - Quora
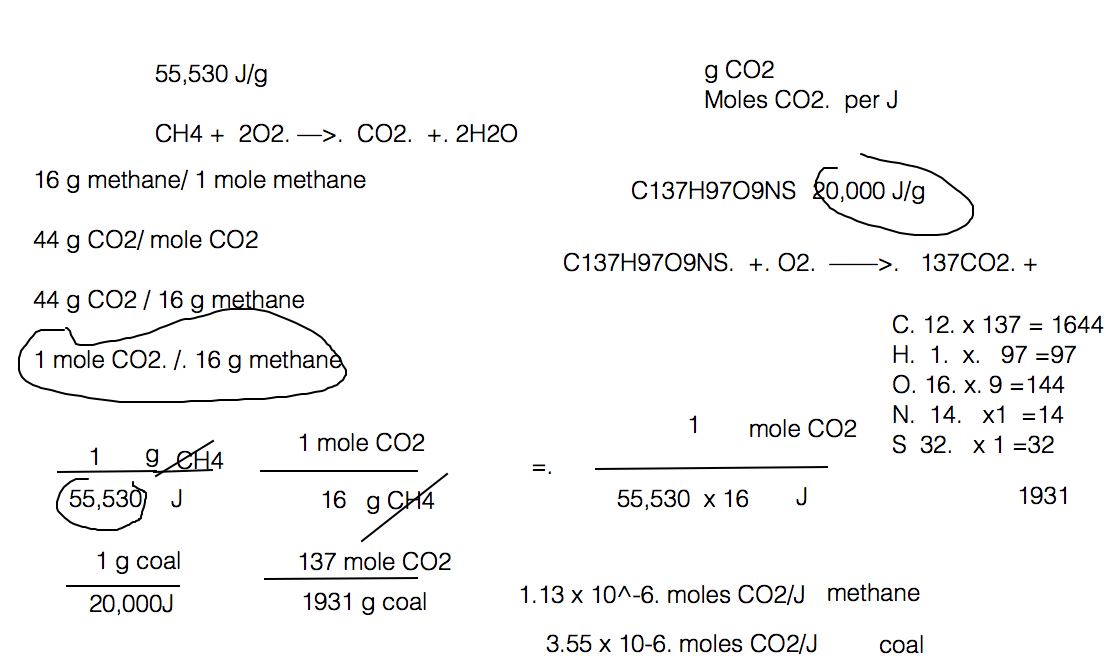
Solved Calculate how many moles of CO2 are produced by
What is the mass of ethene (C2H4) required to burn completely with 40g of oxygen (C=12, H=1)? - Quora
What is the volume of gas present in 22g of carbon dioxide at RTP? - Quora