By A Mystery Man Writer

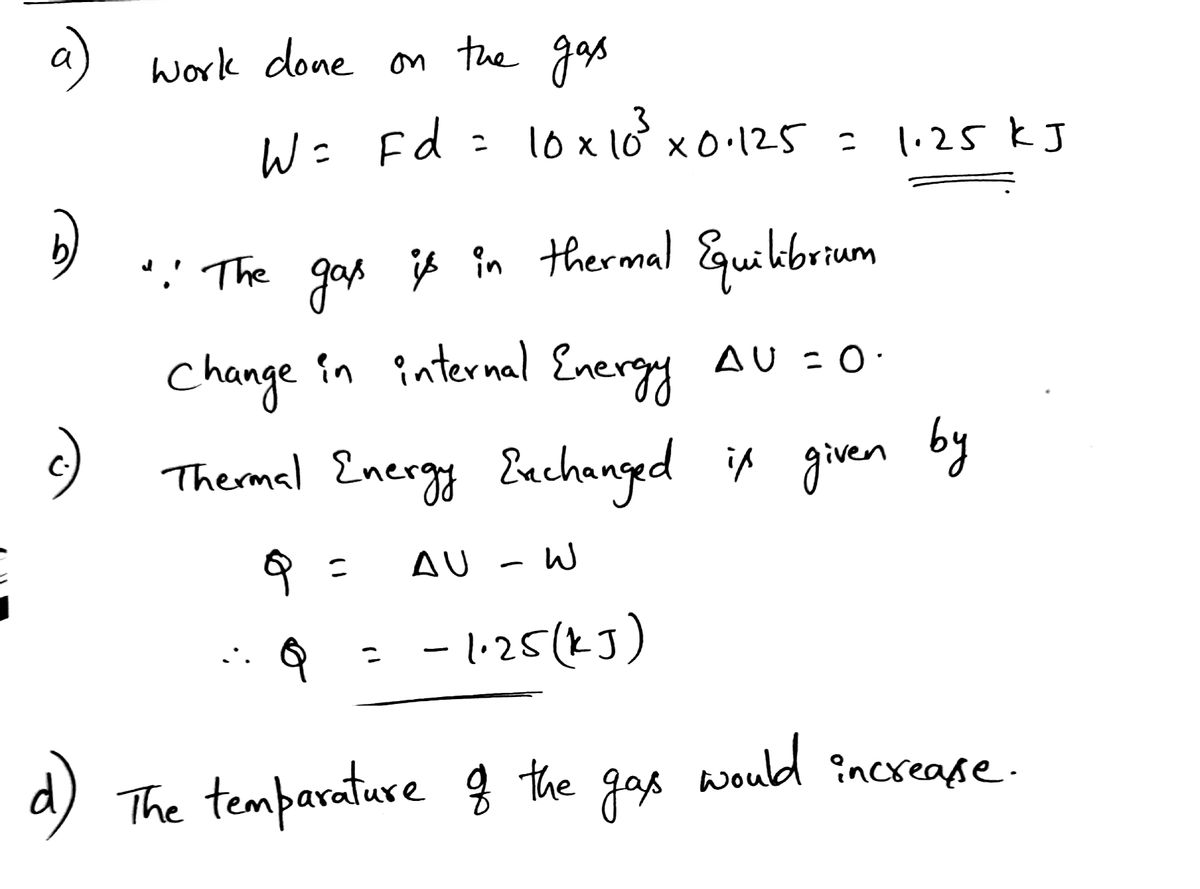
Answered: An ideal gas is compressed from a…

Analysis of three scientific laws using the tool of Fig. 1
Compressed Air - Pipe Line Pressure Loss - Online Calculator with Metric and Imperial Units
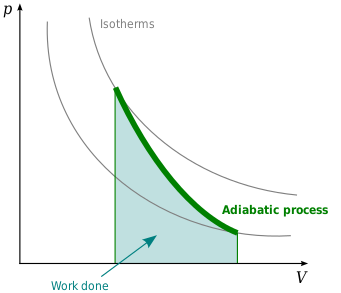
Adiabatic process - Wikipedia

the constant external pressure required to compress 1 mole of an ideal gas from 23 10^-3m^3to 8 10^-3m^3 when work - Chemistry - Thermodynamics - 16657061
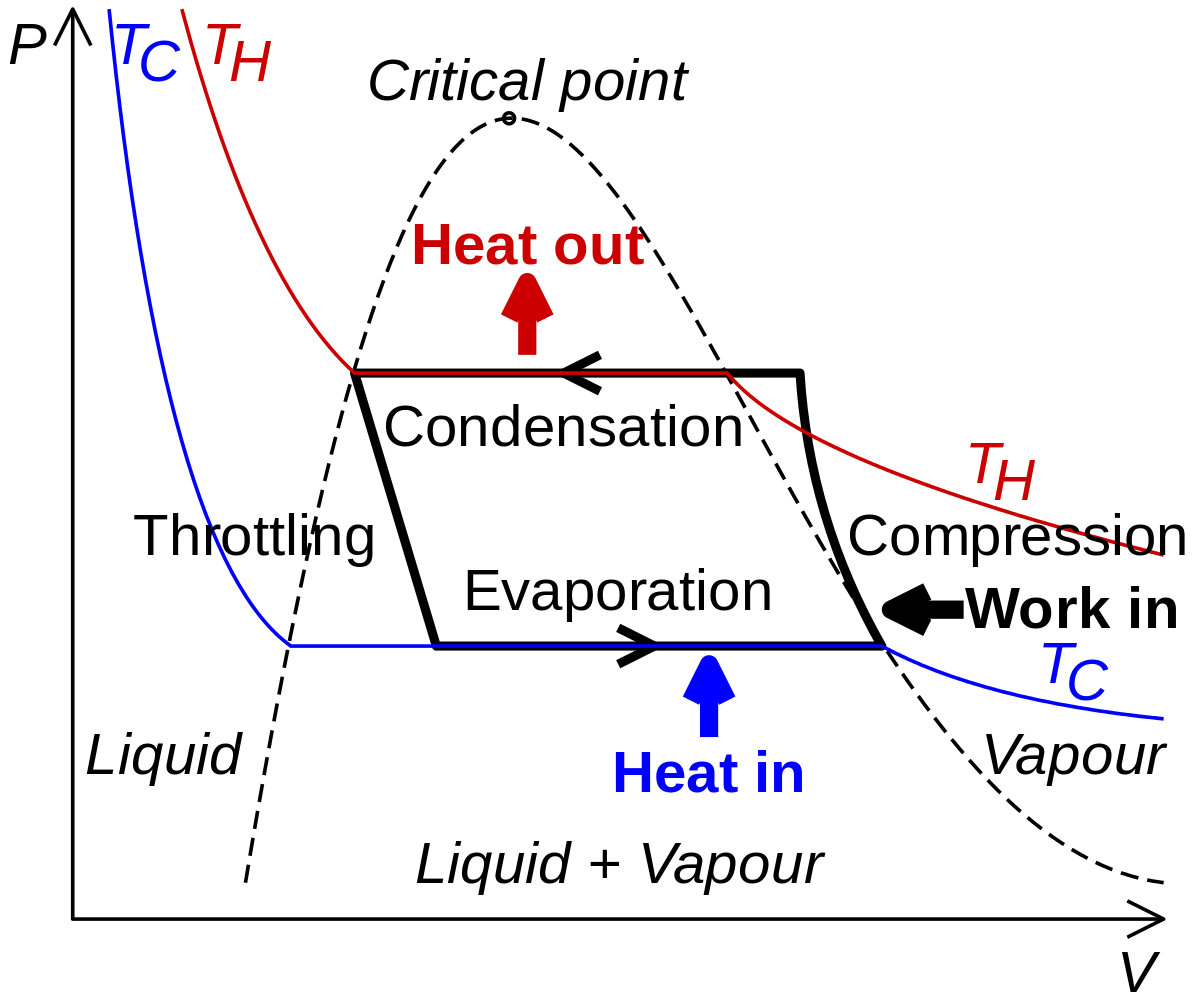
Vapor-compression refrigeration - Wikipedia

Please Pick Up Ice, Water, Steam Quiz Internal Energy, Heat & Work problem Set. - ppt download

Distribution of general chemistry textbook illustrations according

The work done in adiabatic compression of `2` mole of an ideal monoatomic gas by constant