By A Mystery Man Writer

Click here:point_up_2:to get an answer to your question :writing_hand:the compressibility factor z at a lowpressure range of all gases except hydrogen is
Click here👆to get an answer to your question ✍️ The compressibility factor Z a low-pressure range of all gases except hydrogen is-Z-1- displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-Pb-RT-Z - - 1 - displaystylefrac-Pb-RT-
The van der Waals equation for real gases is -P-aVm2-Vm-x2212-b-RT

Compressibility Factor (Z-Factor) Equation of State

2. U 0.52, 0.68, 0.74 At low pressure, the comprensibility factor is given as (1) - RTV Pb RT 12 12 Photo (3) 1+ TV Pb RT 3. 10 mole of an ideal gas 27°C ernands

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be
How can a gas be ideal at a high pressure and low temperature? - Quora
At Critical Temperature,pressure and volume . The compressibility Factor (Z) Is
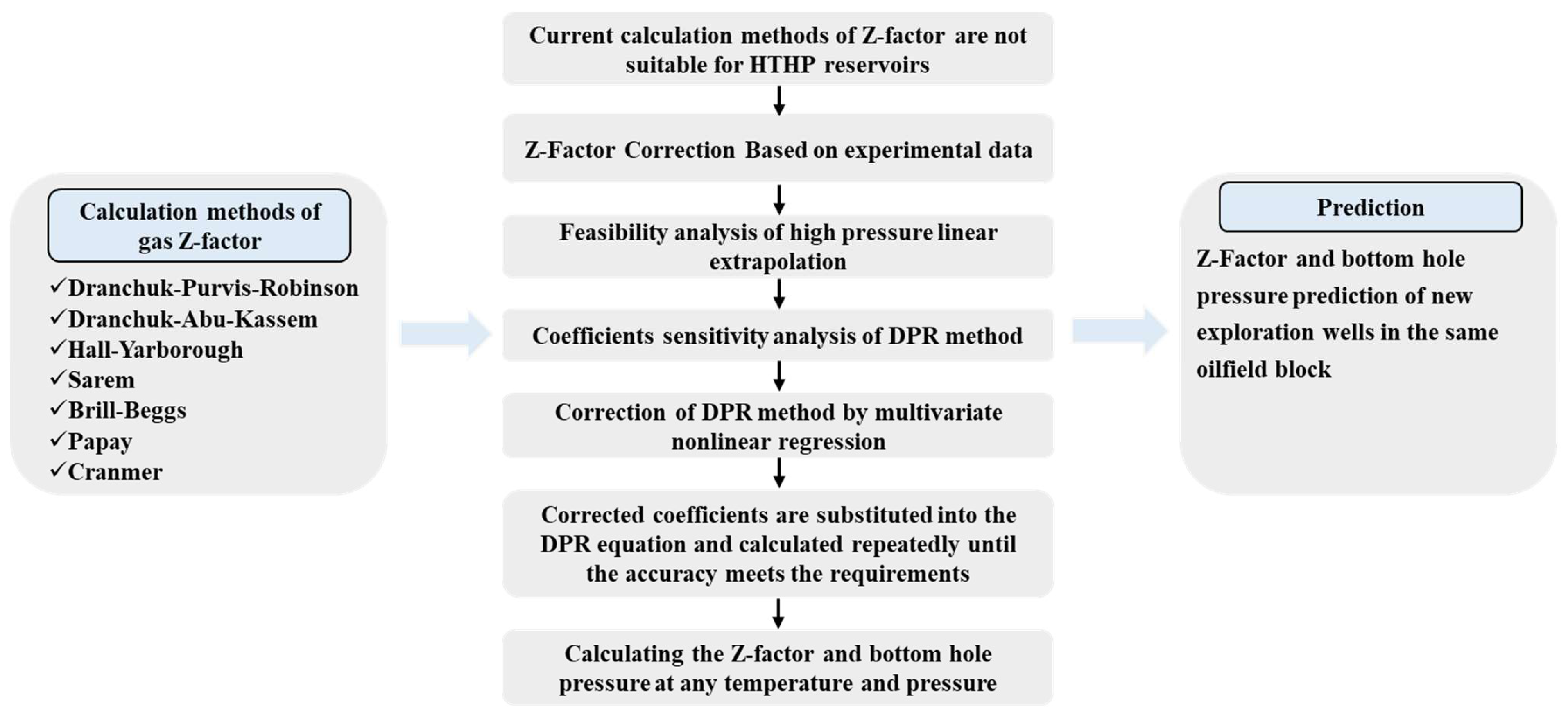
Energies, Free Full-Text

Compressibility factor (z): real gases deviate from ideal behav-Turito

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

Gas compressibility factor Z: Ideal gas vs Real gas

Torateal gas, the compressibility factor Z has different whues different temperatures and pressures. Which of the following is not correct under the given conditions? (a) Z<1 very low pressure. (b) Z>1 high

Except H(2) and He, the compressibility factor Z(=(PV)/(nRT))lt1 for a