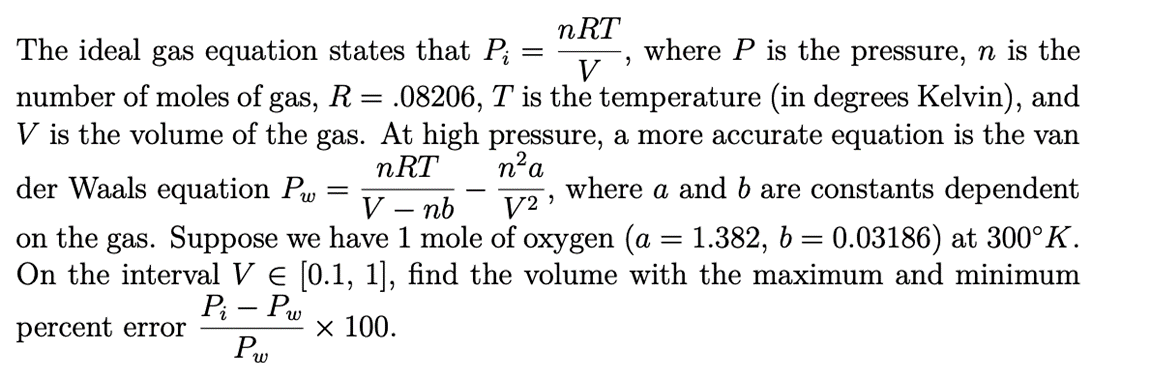
amp;#160;NRT The Ideal Gas Equation States That Pi Where P Is The Pressure, N Is The V Number Of Moles Of Gas, R= .08206, T Is The Temperature (In Degrees Kelvin), And V Is The Volume Of The Gas. At High Pressure, A More Accurate Equation Is The Van NRT
1st law

An ideal gas initially at a state __(P1,V1)__ is allowed to expand isothermally to a state __(P2, V2)__. Then the gas is compressed adiabatically to its initial volume __V1__. Let the final

Equation of State Ideal-gas Equation Where: P = pressure (Pa

Ideal Gas Law: Overview, Questions, Easy Tricks, Rules, Preparation

What is n in the equation of the ideal gas law?
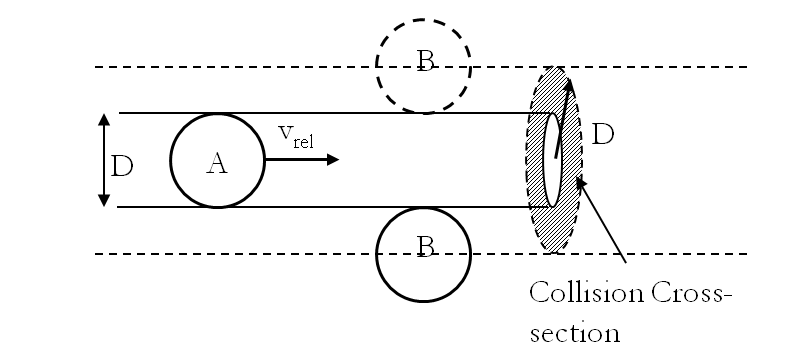
Mean Free Path

Osmosis and Osmotic Pressure: Definition, Formulas, Proof, Videos, Q&A

Giancoli 7th Edition, Chapter 13, Problem 36
What is the equation for calculating the pressure exerted by an

Equation Of State (Ideal Gas), Glenn Research Center
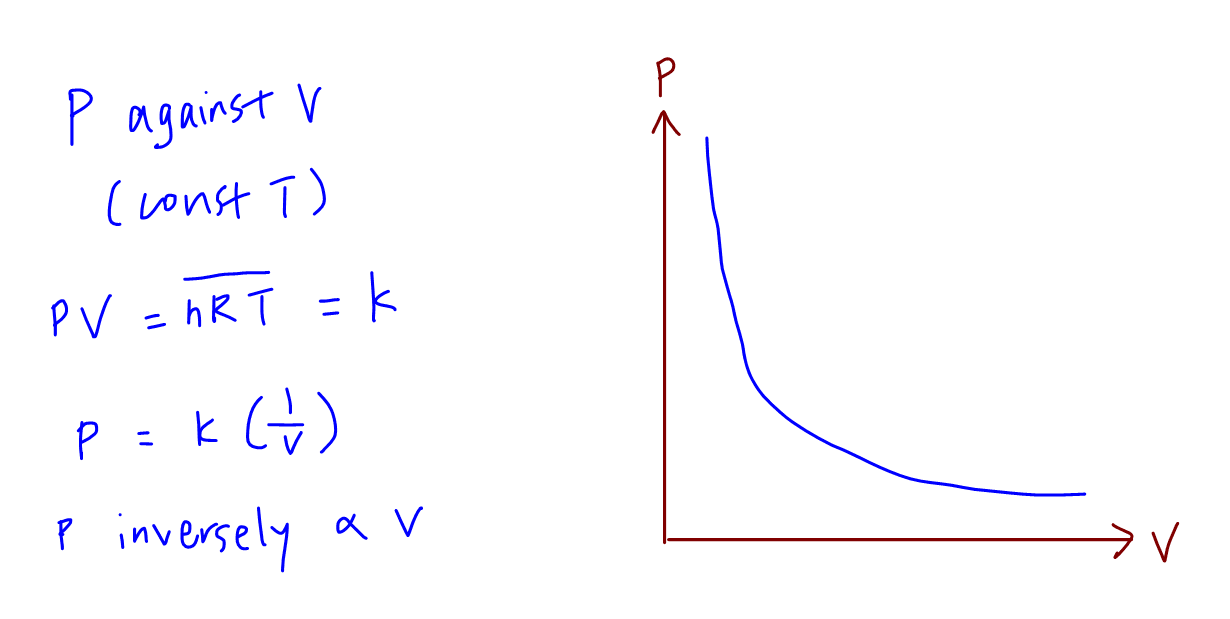
Ideal Gas Graph Sketching