By A Mystery Man Writer
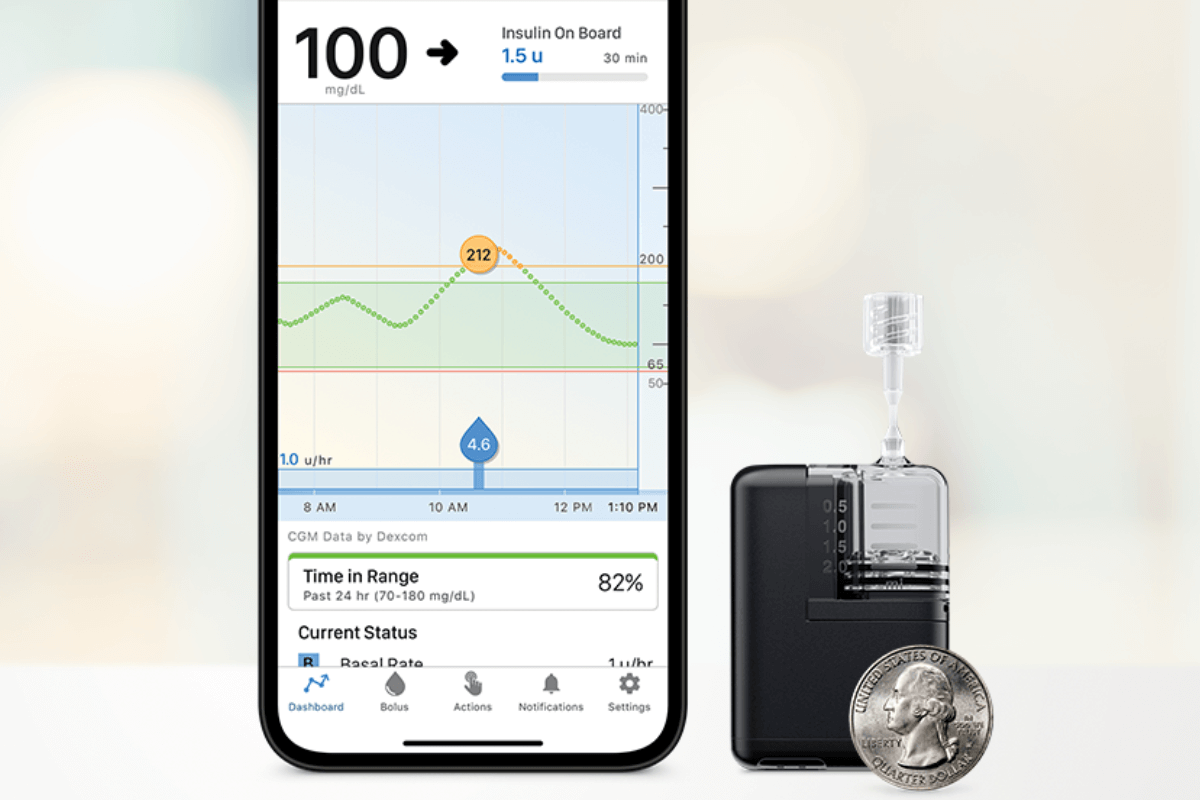
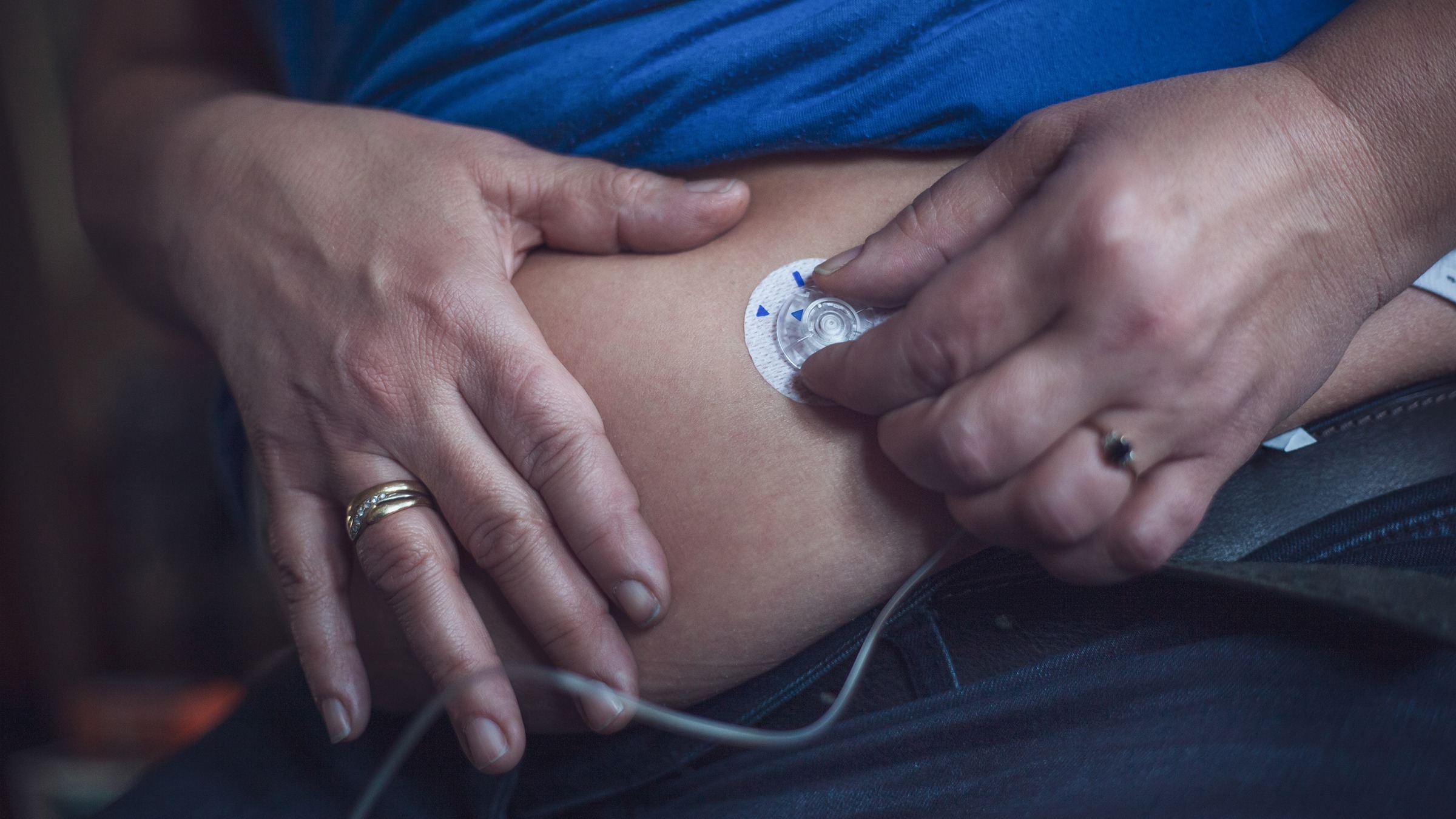
In just the past year, we have had multiple artificial pancreas systems authorized by the Food and Drug Administration (FDA)…and it’s not stopping! Last week, Tandem Mobi—a miniature-sized insulin pump, for use with Tandem’s Control-IQ™ technology and a compatible continuous glucose monitor (CGM)—received FDA clearance. The Tandem Mobi is half the size of the company’s…

JDRF Diabetes Blog & Resources

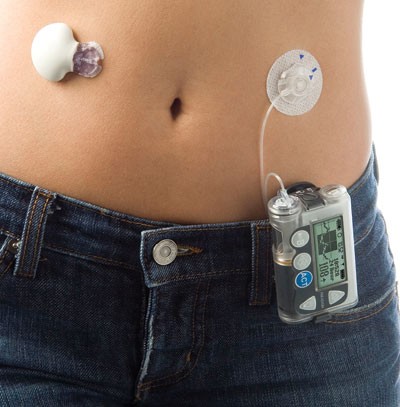
Wearable Artificial Pancreas Device Technology
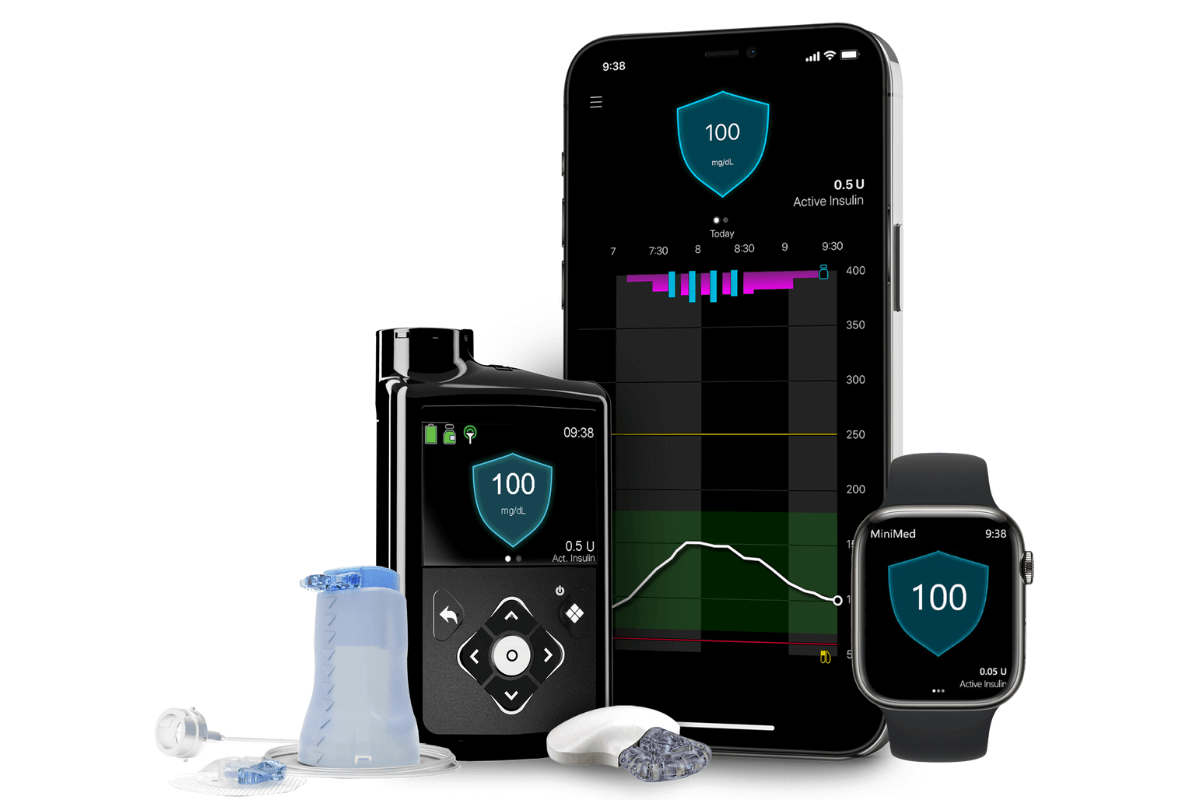
FDA Approves Medtronic 780G Artificial Pancreas System
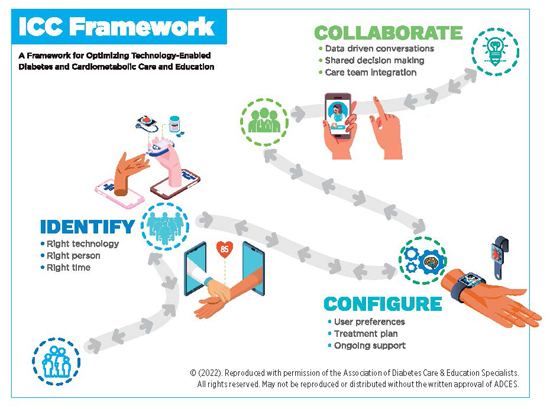
Insulin Delivery Device Technology — Today and Into the Future

Hypoglycemia prevention: PID-type controller adaptation for

Glucose-responsive insulin delivery for type 1 diabetes: The
Medical devices: Managed by machine

Medical devices, smart drug delivery, wearables and technology for

FDA approves first artificial pancreas for type 1 diabetes, Medtronic's MiniMed 670G - CBS News

FDA Clears iLet Bionic Pancreas System for Type 1 Diabetes
Insulin Pumps: Types and Advantages - GoodRx