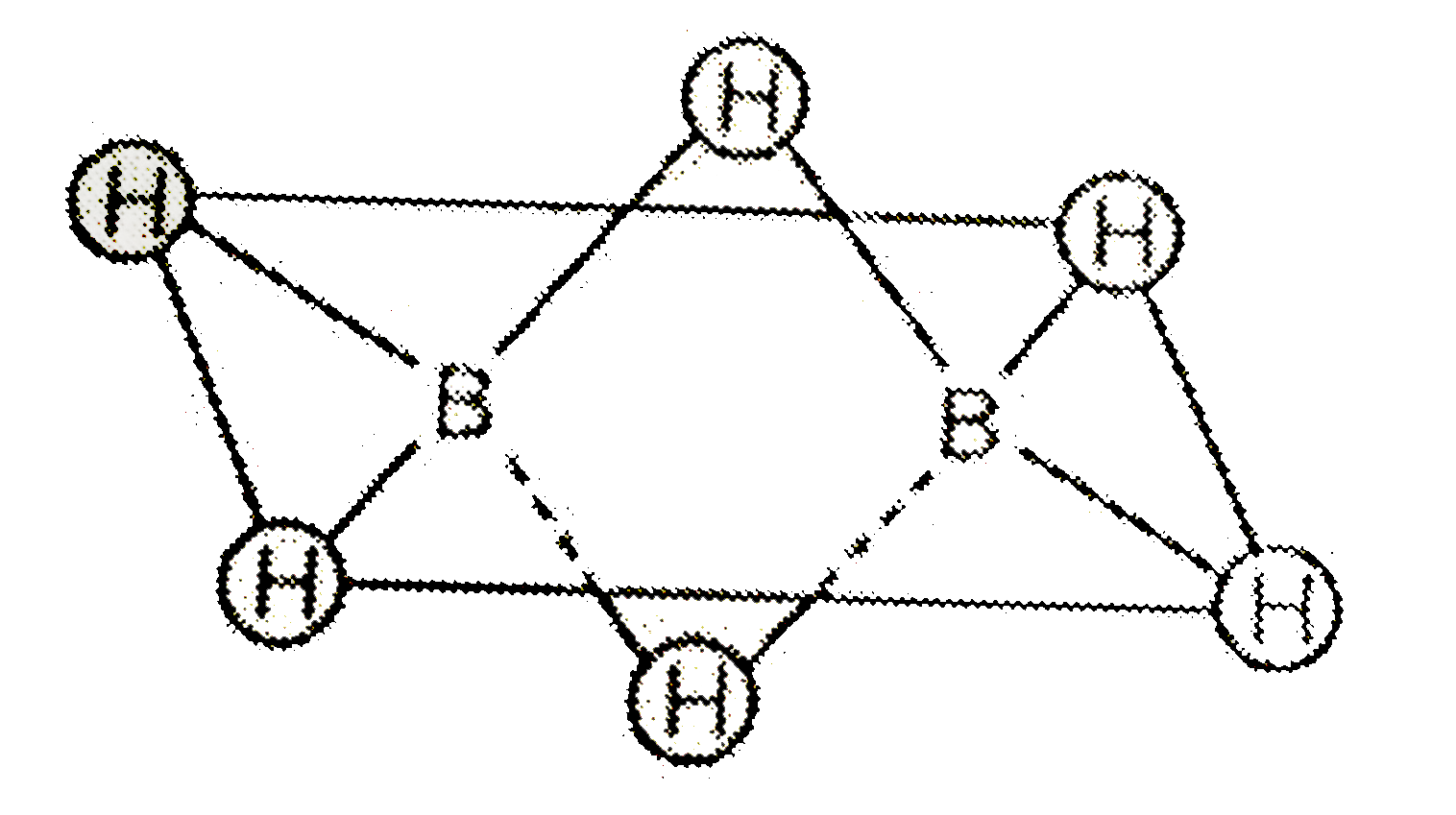
24. Assertion :In B2H6, the terminal B H bonds are shorter, than the B H bridge bonds Reason: The terminal B H bond order is greater than that of the B H bridge bond
24- Assertion-In B2H6- the terminal B-H bonds are shorter- than the B-H bridge bonds Reason- The terminal B-H bond order is greater than that of the B-H bridge bond

B2H6 bonding
Telugu] In the strucute of B(2)H(6), the number of BH(2) groups prese
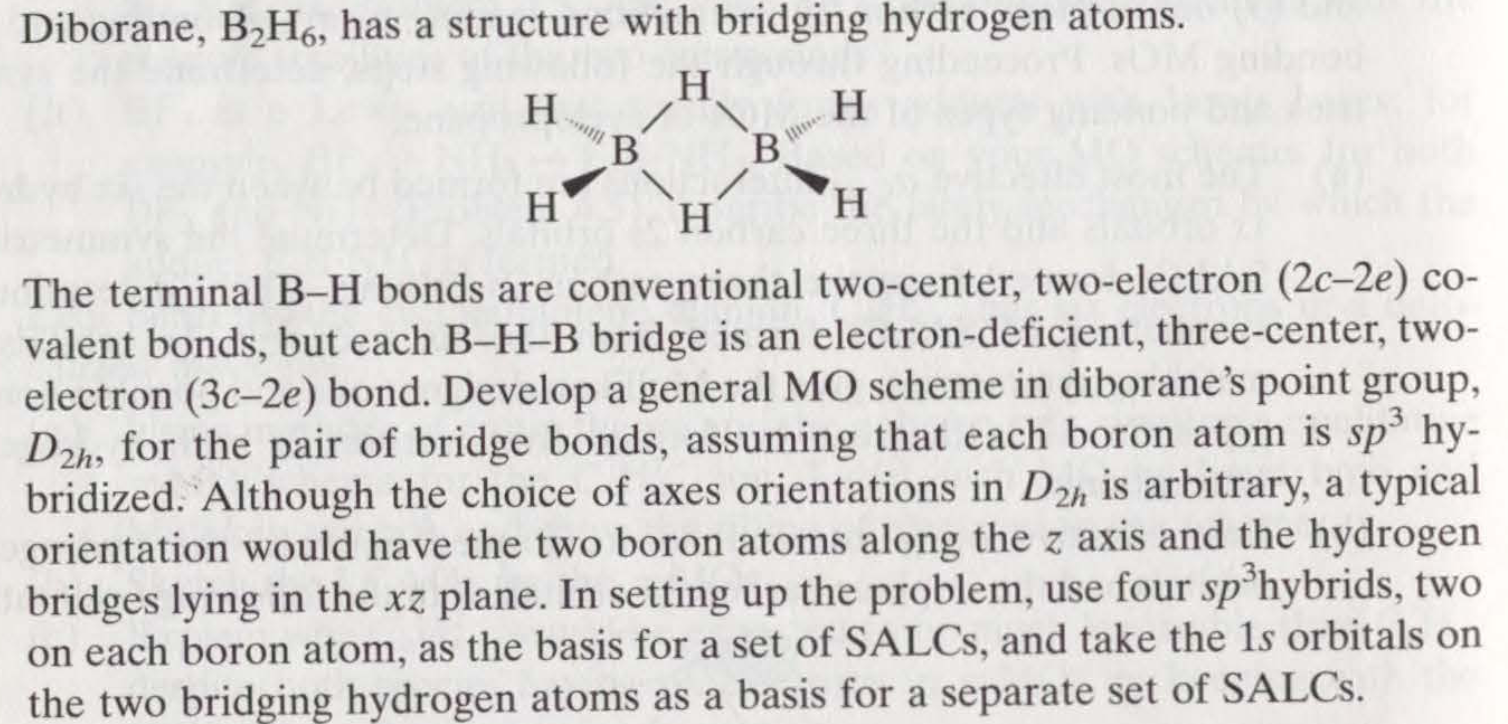
Diborane, B2H6, has a structure with bridging

A new look at the nido-undecaborate system - ScienceDirect

Mechanistic Investigation of Stoichiometric Alkyne Insertion into Pt−B Bonds and Related Chemistry Bearing on the Catalytic Diborylation of Alkynes Mediated by Platinum(II) Diboryl Complexes

How many structures are correct from belowi In diborane 12 valence e are involved in bondingii In diborane, two boron and four terminal hydrogen, lie in the same plane.iii Diborane has ethane

Chemical Bonding (Advance) : Chemistry, PDF, Chemical Bond
How are the B-H-B bridge bonds formed in B2H6? - Quora

Regeneration of ammonia borane by reduction of polyborazylene (PB) by

Assertion: In diborane, each B atom is sp^3 hybridised.Reason: In diborane, the terminal 2-centre-2-electron B-H bonds are called banana bonds.Select the correct option.Both A and R are true and R is the

ChemicalBondingBYPMS PDF

Formation and Reactivity of Electron‐Precise B−B Single and Multiple Bonds - Arrowsmith - 2017 - Angewandte Chemie International Edition - Wiley Online Library