By A Mystery Man Writer

A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU'S to understand more about it.
Standard solution

Primary standard sodium carbonate (Na_2CO_3) was used to

HCL Standardization
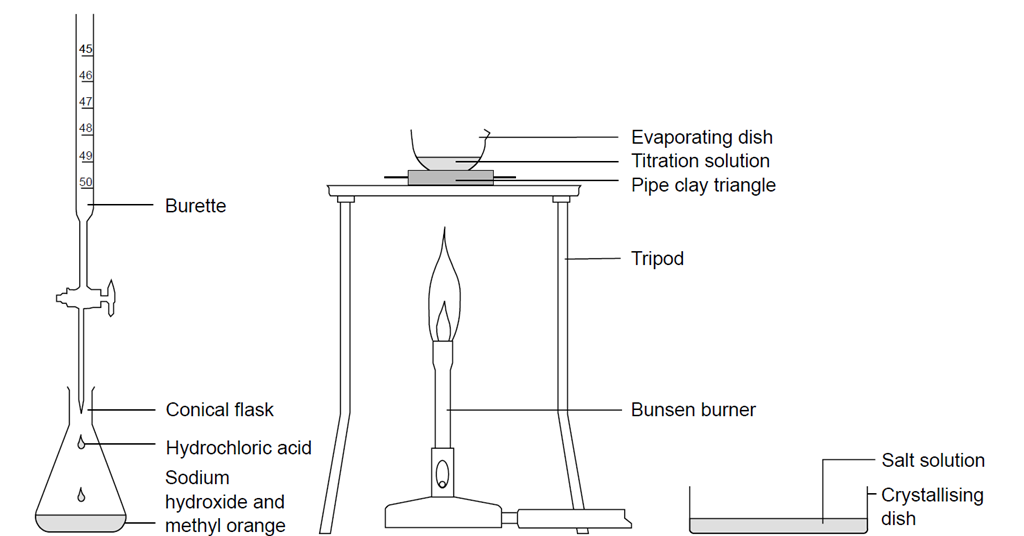
Titrating sodium hydroxide with hydrochloric acid
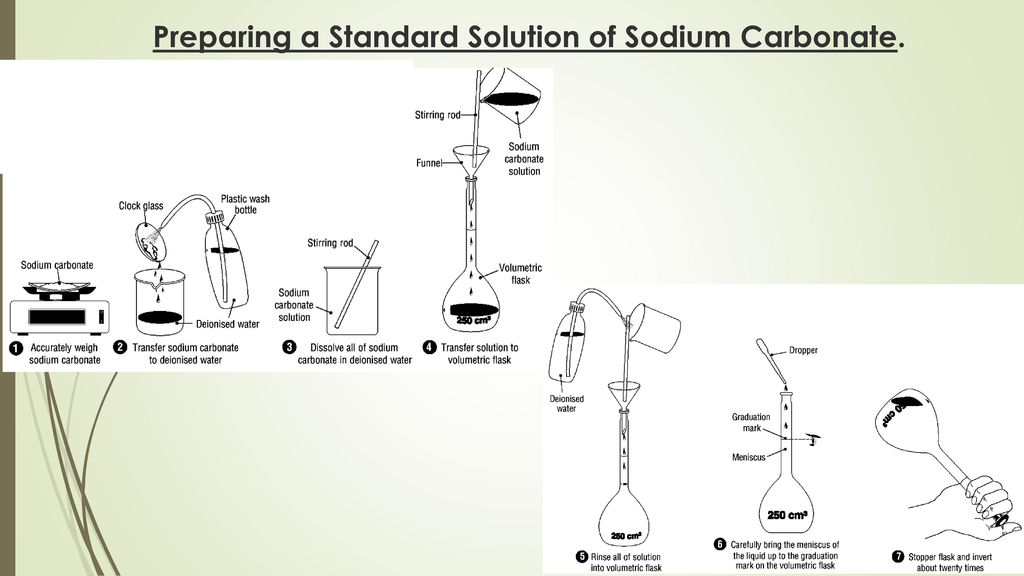
Volumetric Analysis Chapter ppt download

Titration of Sodium Carbonate With Hydrochloric Acid

6. Preparing Standard Acid and Base

LAB Eport titration - Objectives 1. To learn how to prepare a

Carbon Standard, (Sodium Carbonate-Sodium Bicarbonate), Certified

SOLUTION: Preparation of Sodium Carbonate by Solvay's Process

To Prepare A Standard Solution of Sodium Carbonate