By A Mystery Man Writer
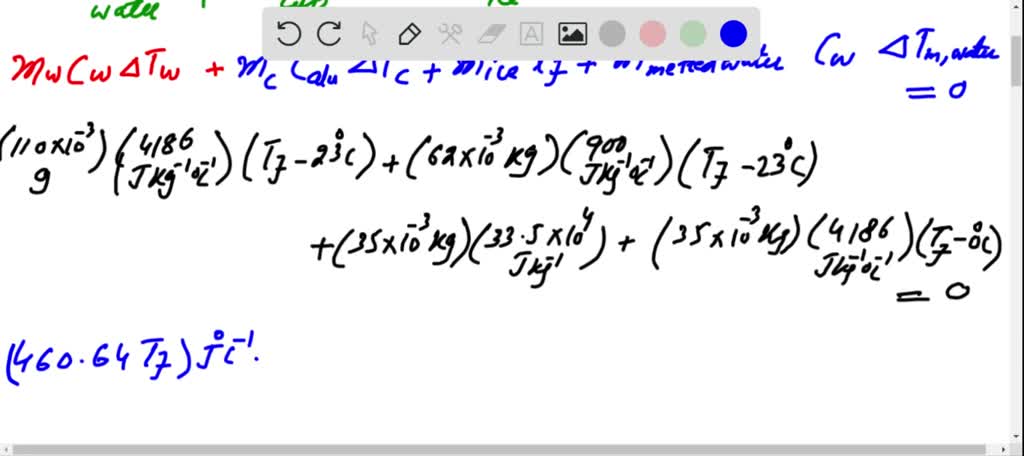
VIDEO ANSWER: let the if is the temperature at thermal equilibrium. Then, by using love, congressional energy became right. He released way water but last year released Why Let me go! Less heat absolved way eyes is
Predict/Calculate A 35- g ice cube at 0.0^∘ C is added to 110 g of water in a 62 -g aluminum cup. The cup and the water have an initial temperature of 23^∘ C (a) Find the equilibrium temperature of the cup and its contents. (b) Suppose the aluminum cup is replaced with one of equal mass made from silver. Is the equilibrium temperature with the silver cup greater than, less than, or the same as with the aluminum cup? Explain.
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.

Answered: A 10 g ice cube, initially at 0 ºC, is…

Solved An ice cube at an unknown temperature is added to
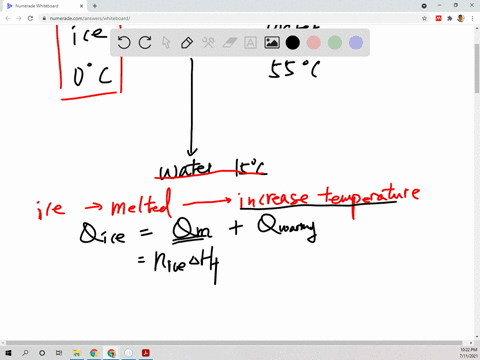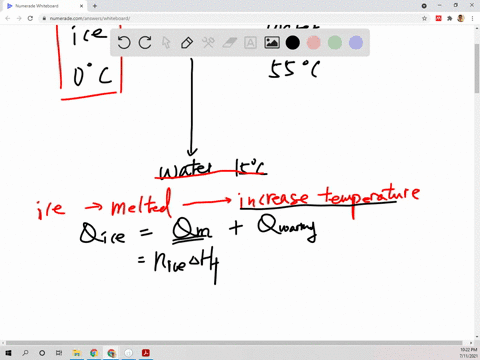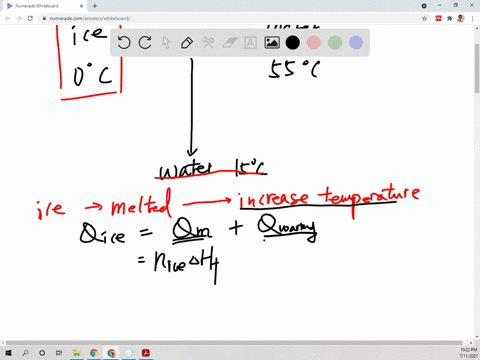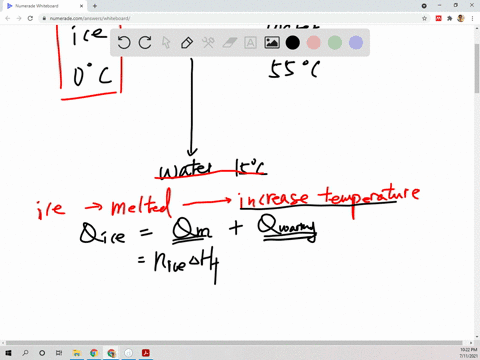
⏩SOLVED:Suppose 45.0 g of water at 85^∘ C is added to 105.0 g of…

⏩SOLVED:Predict/Calculate A 35- g ice cube at 0.0^∘ C is added to…

⏩SOLVED:lt takes 6.01 kJ to melt 1 mol of ice at 0 ^∘ C . Based on…

⏩SOLVED:Predict/Calculate Referring to the previous problem, suppose…

19-20) A 35-g ice cube at its melting point is dropped into an insulated container of liquid nitrog
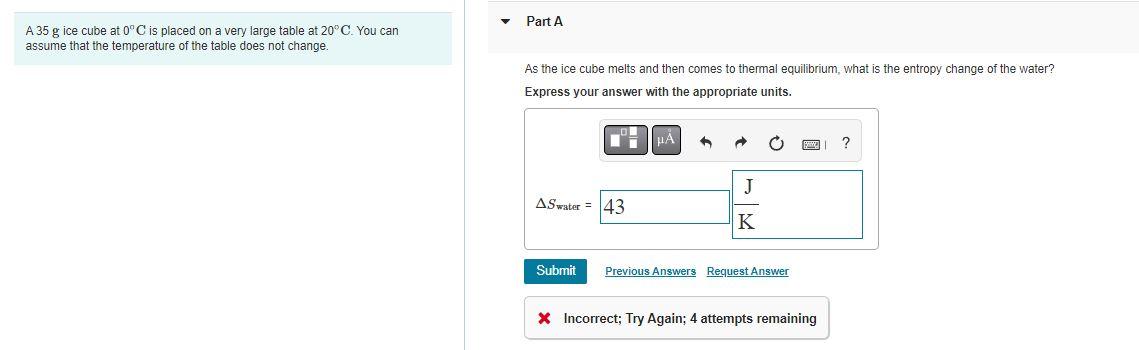
Solved A 35 g ice cube at 0°C is placed on a very large

14.24 A 0.0500-kg ice cube at −30.0ºC is placed in 0.400 kg of 35.0ºC water in a very